Metritis is a common postpartum uterine disease affecting approximately 25% of dairy cows in the United States (Pinedo et al., 2020), and remains a major challenge for herd health and dairy sustainability. Its impacts extend well beyond the initial clinical episode. Affected cows produce less milk (Figueiredo et al., 2024), may incur additional losses due to treatment-related milk withdrawal, and experience compromised welfare (Barragan et al., 2018), increased risk of culling (de Oliveira et al., 2020), and reduced reproductive performance (Figueiredo et al., 2024). More recently, metritis has also been associated with an increased environmental footprint per unit of milk produced, reflecting reduced biological efficiency (Martelo-Pereira et al., under review at Journal of Dairy Science). Collectively, these effects translate into substantial economic losses, estimated at approximately $1.2 billion annually in the U.S. and up to $5.0 billion globally (Pérez-Báez et al., 2021; Rasmussen et al., 2024), underscoring the importance of effective management.
In conventional (non-organic) systems, antimicrobial therapy remains the most consistently effective treatment, increasing clinical cure rates by approximately 30 percentage points compared with no treatment (Chenault et al., 2004; McLaughlin et al., 2012; Lima et al., 2014; Haimerl et al., 2017; de Oliveira et al., 2020; Merenda et al., 2021; Menta et al., 2024). Clinical cure is a critical milestone: cows that fail to resolve metritis produce less milk, take longer to conceive, are more likely to be culled, and incur roughly threefold higher costs than cows that achieve cure ($1,403 vs. $460; Figueiredo et al., 2021; Menta et al., 2024; Martelo-Pereira et al., under review at Journal of Dairy Science). However, even cows that achieve clinical cure do not fully recover reproductive performance to the level of unaffected cows, indicating that resolution of clinical signs does not equate to complete uterine recovery.
This persistent subfertility has directed attention toward underlying uterine mechanisms. Cows with metritis are more likely to develop subsequent uterine diseases, particularly endometritis. Among cows that fail to achieve clinical cure the prevalence of endometritis can exceed 90% (Figueiredo et al., 2021). These conditions can delay uterine involution and further impair fertility. At the tissue level, evidence indicates that metritis induces long-lasting uterine alterations. Cows with a history of metritis exhibit reduced numbers of endometrial glands up to 165 days postpartum (Sellmer-Ramos et al., 2023), increased adenomyosis (Sellmer-Ramos et al., 2025), and persistent endometrial inflammation (Silva et al., 2024; Caldeira et al., 2025; Moraes et al., 2025). Recent work corroborates these findings, showing increased inflammation and periglandular fibrosis at both the histological and transcriptomic level (Rashid et al., 2026; Figure 1). In contrast, the uterine microbiome appears to play a limited role in explaining long-term subfertility, as multiple studies report minimal associations with reproductive outcomes (Figueiredo et al., 2024). These findings suggest that while antimicrobial therapy improves clinical outcomes, it may not address the structural and functional uterine changes that underlie reduced fertility. Supporting this, embryos recovered from cows with a history of metritis are smaller at 15 days and are exposed to lower concentrations of interferon-τ (Ribeiro et al., 2016). Because early embryo development depends on histotroph secreted by endometrial glands, persistent alterations in gland number and function may create a suboptimal uterine environment, thereby reducing embryo viability and increasing pregnancy loss risk.
A recent study of ours did not detect differences in uterine metabolomic profiles between cattle with and without metritis (Rashid et al., 2026), and the mechanisms linking metritis to long-term reproductive inefficiency remain incompletely understood. Further research is needed to clarify how uterine structure, function, and embryo–maternal interactions are altered following metritis and how these changes can be mitigated to improve fertility outcomes.
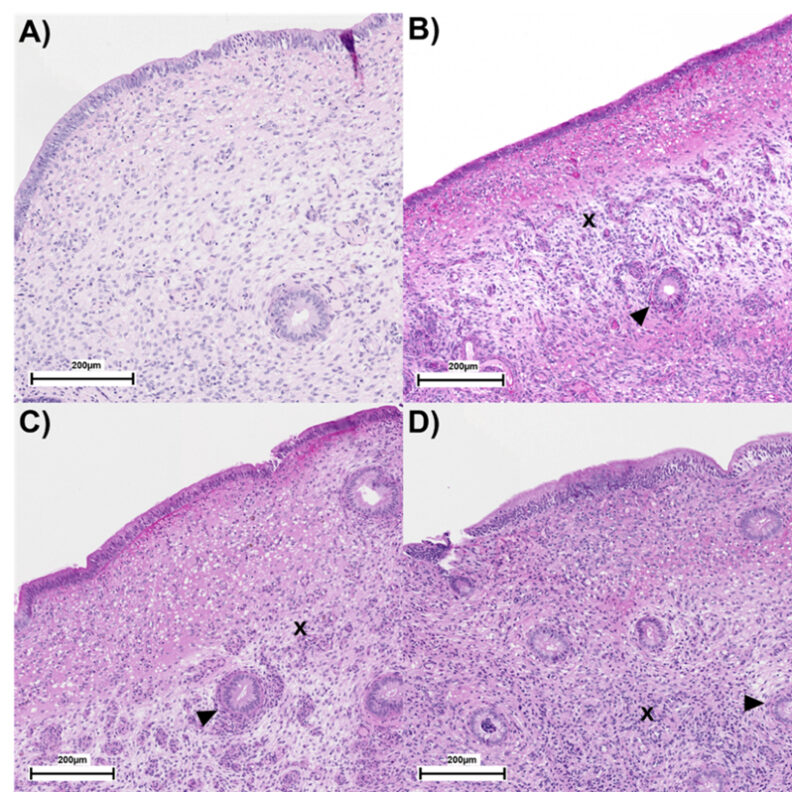
Figure 1. Endometrial biopsies graded using the Kenney-Doig grading system. Grade I (Panel A): normal endometrium or mild, focal inflammation or fibrosis. Grade IIA (Panel B): mild to moderate inflammation and/or multifocal fibrosis. Grade IIB (Panel C): moderate inflammation and/or multifocal to diffuse fibrosis. Grade III (Panel D): severe inflammation and/or diffuse fibrosis. Arrowheads represent peri-glandular fibrosis, whereas x represents increased cellular density (diffused inflammation). Adapted from Assessing Long-Term Changes in Endometrial Morphology and Functionality in Dairy Cows With Metritis Rashid et al. (2026).
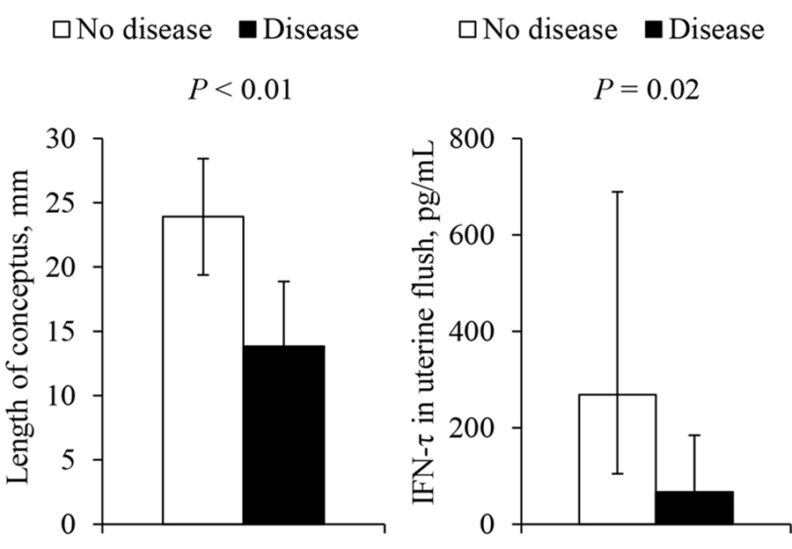
Figure 2. Length of the recovered intact conceptuses, and concentration of IFN-τ in the uterine flush of pregnant cows. Adapted from Ribeiro et al. (2016).
