By Caio Figueiredo, Veterinary Medicine Extension
We just returned from Kentucky, where the American Dairy Science Association’s Annual Meeting was held. As always, the meeting was great and it was nice to catch up with friends and talk about dairy cows, in addition to learning more about our field. I was particularly impressed with new developments related to mastitis management that were presented during the lactation biology symposium and the udder health sessions. Shortly after landing in Pullman, my thought was: “Why not highlight in the summer Extension newsletter a few of the papers and abstracts published in 2025?”. Well, here we are!
Among the many publications related to mastitis, I decided to focus on recent updates regarding diagnostic and monitoring tools, and possible future treatment strategies for mastitis in dairy cows. Historically, somatic cell count (SCC) has been widely used as an indicator of udder inflammation and recently, differential somatic cell count (DSCC) has become available as an auxiliary tool for milk quality control, with the potential to indicate different stages of inflammation when combined with SCC. A study by Fonseca et al. (2025) aimed to assess the association of SCC and DSCC with milk yield and milk components at the quarter level, using a bi-weekly convenience sample from 5 dairy herds that implemented automated milking systems (AMS). The results showed that elevated SCC, in combination with lower proportions of DSCC, was associated with the highest milk loss for primiparous and multiparous cows. For example, the estimated milk loss for a quarter with a SCS of 7 and a DSCC of 55% was 1.45 kg/d as compared to a quarter with a SCS of 2 and DSCC of 65% in multiparous cows. The association was similar when the outcome was the lactose content. Quarters with elevated SCS and lower DSCC had the lowest lactose percentage. No notable changes in fat content were observed across different SCS levels in multiparous cows, and quarters with higher DSCC had the lowest fat percentage in primiparous and multiparous cows. Protein content tended to be lower in quarters with increased SCS and low DSCC. In quarters from primiparous cows with DSCC levels above 70%, protein content showed slight variation across SCS levels. For multiparous cows, however, protein content remained relatively stable across different SCS and DSCC levels. Overall, the combination of elevated SCS and low DSCC was associated with the most substantial milk loss, highlighting the potential use of such a diagnostic tool to optimize udder health management.
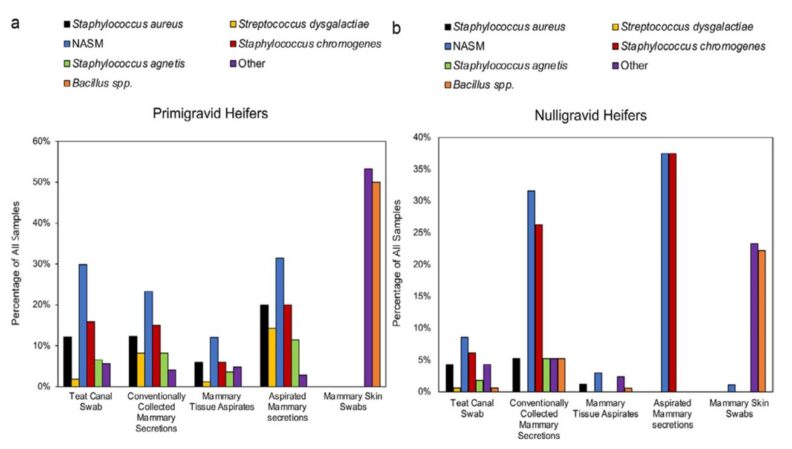
Another publication that caught my attention was generated by researchers at the University of Missouri. Novo et al. (2025), assessed several aspects associated with intramammary infections (IMI) in dairy heifers, and the use of different diagnostic tools including fine needle aspiration. In their study, 304 quarters were sampled from 152 Holstein dairy heifers between the ages of 6 to 24 mo. A more rigorous definition of IMI in nulliparous heifers, hIMI, was used to quantify hIMI occurrence. This definition is comprehensive and primarily defined by the culture results of conventionally collected mammary secretions and aspirated mammary secretions or mammary tissue aspirates. Teat canal swabs and mammary skin swabs served as secondary sample types to minimize the influence of skin and teat canal flora on the results of primary sample types. Overall, 304 teat canal swabs, 93 conventionally collected mammary secretions, 304 mammary skin swabs, 43 aspirated mammary secretions, and 257 mammary tissue aspirate samples were collected, of which 24.7% (75), 39.8% (37), 34.5% (105), 48.9% (23), and 10.1% (26) were culture positive, respectively (Figure 1). The most common bacteria identified among all sample types, except mammary skin swabs, were staphylococci (primarily non-aureus staphylococcal and mammaliiococcal species) from both primigravid and nulligravid heifers. The overall hIMI occurrence was 28.5% (43/151) of heifers and 18.1% (54/298) of quarters. The highest proportion of quarter-level infections was seen in the mid-gestation group (42.5%, 17/40), followed by the late gestation group (35.7%, 10/28) and the early gestation group (23.1%, 12/52). The mature (11–16 mo) nulligravid heifers and young (6–10 mo) nulligravid heifers had similar quarter-level hIMI occurrence of 7.8% (7/90) and 9.1% (8/88), respectively. Overall, this study highlighted when hIMI occurs and the bacterial genera and species involved, and provided results based on specific sampling methods that may prove useful for future studies evaluating hIMI status. At ADSA, multiple abstracts related to diagnostic tools for mastitis were presented, among them abstract #1194 by Coffman et al. commented on the use of infrared thermal imaging as a possible real-time diagnostic tool for mastitis detection in the future.
Mastitis is the most prevalent disease in dairy cattle and is the major driver for antimicrobial use in the dairy industry. A lot of research efforts have been devoted to developing therapeutic strategies for mastitis that reduce the use of antimicrobials or at least improve antimicrobial treatment efficacy. A research group from Europe assessed the use of a nonsteroidal anti-inflammatory drug (NSAID) as an initial treatment for non-severe mastitis. Krömker et al. (2025) conducted a randomized, multi-herd, noninferiority study to compare 2 treatments for non-severe mastitis cases. The animals in the studied group were treated with a systemic NSAID (ketoprofen; KE group), followed by antibiotic (AB) treatment only in cases without clinical improvement, and the animals in the reference group (AB group) received an intramammary AB. The study included 222 cases of non-severe clinical mastitis on 3 conventional dairy farms in northern Germany between November 2022 and November 2023. Clinical cure (84.7% and 61.5%) and bacteriological cure (79.3% and 61.2%) were significantly higher in the AB group than in the KE group. The risks for recurrent cases (7.3% and 15.7%) were significantly lower in the AB group than in the KE group. The new infection risk did not differ significantly between the treatment groups (6.8% in the AB group and 6.7% in the KE group). In 87% of the cases of the KE group, a subsequent antibiotic treatment was not necessary to reach clinical cure on d 5. The use of KE as the sole initial treatment for non-severe mastitis led to a reduction of antibiotic doses by 86%. Their findings revealed that systemic treatment with KE resulted in lower clinical and bacteriological cure rates, higher recurrence rates of clinical mastitis, and an elevated risk of new infections compared with the antibiotic-treated group. Nevertheless, in more than 85% of cases treated with KE, additional intramammary antibiotic treatment was unnecessary. Thus, this concept represents an alternative mastitis treatment that promotes the deliberate and selective use of antibiotics but has limitations in terms of treatment efficacy.
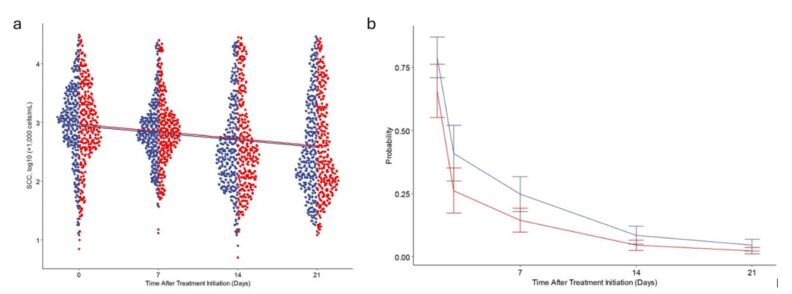
Finally, a recent study from New Zealand (Munn and McDougall, 2025), assessed the effects of the addition of the corticosteroid hydrocortisone aceponate (HCA) to a procaine penicillin G intramammary infusion on clinical outcomes and bacteriological cure rates of naturally occurring bovine mastitis. Cows with clinical mastitis were randomly assigned within farm to be treated with penicillin and HCA (IVP) or penicillin alone (CP) on 3 occasions at 24-h intervals following collection of a milk sample. Cows were enrolled over 2 yr: 65 quarters (59 cows) in yr 1, and 66 quarters (61 cows) in yr 2. Cows with subclinical mastitis were enrolled if they had a SCC > 200,000 cells/mL at the most recent DHI test and had no signs of clinical mastitis. Following bacterial isolation from a milk sample, quarters were enrolled and treated. Streptococcus uberis was the most common isolate before treatment in cows with clinical mastitis, and Corynebacterium spp. and NAS the most common outcomes in cows with subclinical mastitis. Bacteriological cure was defined as non-isolation of the bacterial species present pre-treatment from any sample post-treatment. The bacteriological cure rate did not differ between treatment groups in the final multivariate model in the clinical or subclinical arm. The bacteriological cure rate of IVP was non-inferior to that of CP in the subclinical arm, and treatment at 24-h intervals was non-inferior to treatment at 12-h intervals. In the clinical arm, post-treatment SCC were lower for quarters in the IVP than the CP group 14, 21, and 28 d after initiation of treatment. Quarter-level SCC did not differ between treatment groups in subclinically infected quarters. For clinical cases, the proportion of quarters with heat or swelling was reduced following treatment with IVP compared with CP. It was concluded that the bacteriological cure rate of subclinical mastitis was non-inferior following treatment with penicillin and HCA compared with penicillin alone and for 24-h compared with 12-h treatment intervals. There was evidence of reduced severity of local inflammation in cases of clinical mastitis when HCA was combined with penicillin compared with treatment with penicillin alone. At ADSA, multiple abstracts related to preventative/treatment strategies for mastitis were presented, among them, abstract #1342 by Gao et al. assessed the effect of intramammary infusion of chitosan on immune cell infiltration and mammary health in dairy cows, highlighting the potential use of chitosan to improve udder health.
As we move forward with scientific discoveries to improve the sustainability of the dairy industry, we believe that a crucial component for reaching our overarching goal is communication between dairy industry stakeholders and academia. With that in mind, we are happy to share that Dr. Luciana B. da Costa, Dairy Extension Veterinarian from The Ohio State University, will participate in our WSU Vet Med Extension Webinar Series covering several aspects related to pathogens that cause mastitis in dairy cows on October 1st at 4 PM (Pacific Time).