Written by: Dale A. Moore, Katy Heaton, Sandy Poisson, and William M Sischo
The calf’s environment includes feed and water, materials and equipment used for handling and distributing feed and water, materials and equipment used for cleaning, and the physical housing environment which provides shelter. Some attributes of the environment directly influence calf health (such as ventilation) and impact behavior (individual vs. group housing), but the environment and how it is managed has a significant influence on the exposure a calf has to disease pathogens. The purpose of this paper is to describe reasons for pathogen loading in the environment and ways to reduce this loading.

The pathogens
The major reason to manage pathogen load within the calf environment is animal health, but it is important to recognize that some calf pathogens have public health implications for employees, their family, and the community. The names of the pathogens are familiar and many are associated with diarrheal disease in pre-weaned calves and include E. coli, Salmonella, Coronavirus, Rotavirus, Campylobacter, Cryptosporidia, and Coccidia. Occasional cases of diarrhea may be due to Giardia spp or Clostridium perfringens and new information provides evidence for the role of Clostridium difficile toxins in calf enteritis (Hammit et al., 2008). Some of these calf pathogens (e.g. Salmonella, Cryptosporidia, Campylobacter) also have public health implications and there are other bacteria that are strictly of public health concern including the shigatoxin-producing E. coli such as O157:H7 which is not associated with disease in calves (Nielsen et al., 2002). Many pathogens can persist in the environment for weeks to years leading to their build up if not properly managed.
A model for environmental loading of pathogens
Although there are numerous studies of prevalence and potential risk factors for calfhood diseases (Lefay et al., 2000; Uga et al., 2000; Veling et al., 2002; Svensson et al., 2003; Lundborg et al., 2005; Trotz-Williams et al., 2007; Uhde et al., 2008; Younis et al., 2009; Gulliksen et al., 2009; Bartels et al., 2010), there is little research on prevalence and how to remediate the load of pathogens in the environment. The qualitative model below summarizes major factors that may
increase (or decrease) the environmental pathogen load. No data describes how much (quantitatively) each of these paths (denoted by the arrows) contributes to the overall load. Such data would provide valuable insights for prioritizing pathogen reduction interventions.
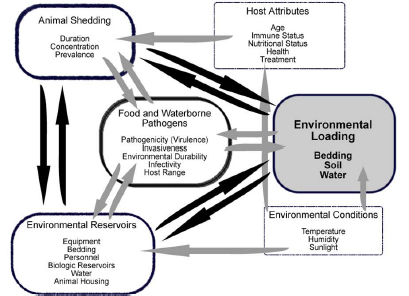
The significance of environmental pathogen loads to dairy calf rearing
Most dairy farms in the US milk year round and newborn calves are continuously being introduced to the farm. Since most farms are designed with fixed and specific housing to accommodate calf management tasks, calf rearing invariably occurs on a specific site. While the physical design allows for a relatively consistent management approach for rearing calves, the challenge in a continuous flow environment is that pathogens introduced into the housing area have the possibility to persist and become endemic (established). Because of constant introduction of new animals and rapid changes in the environment, calves are being raised in disturbed microbial ecosystems. While dynamics of this ecosystem are not well studied, it is certain that the microbial populations are not stable which could mean dramatic fluctuations in pathogen numbers.
Introduction of pathogens to the calf environment
There are two approaches for rearing calves: 1) calves are raised on the dairy of origin, or 2) calves are raised on custom calf raising operations (calf ranches). Calves reared on the dairy of origin are moved from common calving areas to calf housing. Calves are transported to the housing area in a variety of ways, but the active moving of animals implies that potential pathogens are being introduced from one housing area (containing adults) to the calf environment. Calf ranches buy or contract-rear bull and heifer calves usually as day-old calves. Often calves arrive to ranches from multiple sources which may change from day-to-day. Thus, there could be a variety of pathogen types and strains to which calves are exposed (Wray et al., 1990a).
While movement of calves to housing areas is likely a significant source of moving pathogens, pathogens can be introduced into the calf environment from external sources such as equipment, farm personnel (Rice et al., 2003; Kirk et al., 2002a), aerosols, and feed (milk, water, contaminated grain) (lejeune et al., 2001a; Hancock et al., 1998; lejeune et al., 2001b; Davis et al., 2003). In any calf raising system there are many opportunities to introduce potential pathogens into the calf environment. The health of the calf then becomes a function of the pathogen’s ability to survive and possibly amplify in the environment, infect an animal, and cause disease.
Animal sources for environmental loading
It can be argued that there are many ways and sources for pathogens to enter the calf housing environment but it is likely that there is limited amplification of these organisms in the environment. It may also be argued that infected animals are the most likely source for amplification and environmental loading.
One principle of infectious disease is that manifestation of a disease state is a function of the interaction of the host, pathogen, and the environment. Calf diarrhea has a specific pattern of clinical presentation and shedding of agents. The chart below shows that on two different farms, the peak of the proportion of calves with diarrhea occurred in the second week of life.
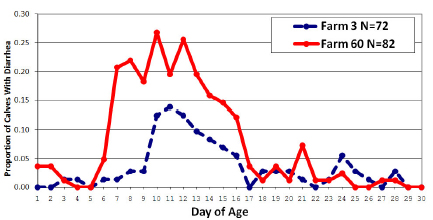
The immune status of the calf and the biology of the agent impact the infection and shedding pattern, but it is likely that these peaks are significantly a function of exposure from the environment. The increase in the proportion of calves with diarrhea reflects an amplification of the pathogen within the animals. Buildup in the environment from amplification creates continuous building and maintaining of an infectious dose that then impacts susceptible calves. Cryptosporidia infection is a good example of amplification and shedding. Susceptible calves are infected with relatively low doses but shed very large numbers of oocysts into the environment (Nydam et al., 2001). That this infection includes host factors as well as environmental contamination is supported by the observation that calves can quickly be detected shedding bacterial pathogens after birth (Hoyle et al., 2004) but do not manifest clinical disease until much later. Essentially, newborn calves shedding pathogens reflect the maternity pen environment and are passing these pathogens through but not amplifying them.
Pathogens can be shed from animals without clinical disease, after clinical disease, and can persist in the environment. Many studies have found that specific Salmonella bacteria are persistent in the absence of any apparent clinical disease for up to 2 years on some premises (McLaren & Wray, 1991; Gay & Hunsaker, 1993). Following clinical disease, the bacteria may be shed for 30 to 50 days (Alexander et al., 2009; Cummings et al., 2009). The table below summarizes some information on shedding and persistence of different pathogens.
| Pathogen | Duration of shedding | Persistence in the environment |
|---|---|---|
| E. coli | 2 days | up to 3 months |
| Rota/corona viruses | 6 days | up to 1 year |
| Salmonell | 30 to 50 days | up toe 2 years |
| Cryptosporida | 10 days | up to 2 years |
How do we remediate pathogen load? Stopping the cycle
Three areas that impact environmental loading.
Introduction
The main objective is to prevent the introduction of new pathogens into the environment. Minimizing the number of sources from which animals originate and ensuring that equipment used to move animals is kept clean are essential prevention measures. Feeding utensils can serve as fomites upon which infectious agents can be carried from one calf to another. Cleaning these items between uses should be standard procedure, but may not be done on every farm. In a cross-sectional study of 119 dairy herds in Ontario, Canada, controlling for other risk factors, the use of soap or a detergent when washing feeding utensils was protective for the risk of C. parvum oocsyt shedding (Trotz-William et al., 2008). Another way to minimize risk is to maintain biosecurity procedures on the farm by keeping personal and equipment dedicated to calf management rather than moving across the farm. A study by Kirk et al. showed that Salmonella could survive on the bottom of rubber boots an extended period of time and could serve as nice transport “vehicle” (fomite) to move Salmonella around the farm (Kirk et al., 2002b).
Amplification
Amplification of pathogens does not occur to any extent in the environment. Most amplification occurs within the animal and often as a part of the disease process. Since disease manifestation is a function of the interaction of pathogen, host, and environment, managing amplification is managing that interaction. Supporting host immunity may be accomplished through nutrition and, most importantly, through achieving adequate transfer of passive immunity through colostrum. Any measure that allows the host to respond to an infection challenge with minimal clinical manifestation will likely minimize amplification. Minimizing pathogen spread between animals will reduce the probability of spreading infectious doses of pathogens from infected to susceptible animals. One method to accomplish this is through the use of individual hutches during the most susceptible times for a calf to manifest clinical infection.
Loading
The concept of developing interventions on the farm to decrease the environmental load of food and water-borne pathogens originating from animal agriculture is relatively new. Smith et al. demonstrated a beef calving system aimed at decreasing environmental exposure of calves to neonatal diarrheal-associated pathogens (Salmonella and Cryptosporidium) (Smith et al., 2003). The aim of the system was to isolate neonates from older calves that are entering or leaving the high risk period for shedding of these pathogens (5 to 20 days of age). The system was effective at reducing the infectious dose in the pasture environment which was associated with decreasing the rates of neonatal diarrhea.
In the dairy calf environment, use of concrete flooring was considered “protective” for calves shedding C. parvum oocysts (Trotz-William et al., 2008) and use of “slatted flooring” was considered a risk factor for diarrhea (Gulliksen et al., 2009). This could be due to the inhospitable environment for the pathogen on solid concrete or could be associated with easier cleaning and disinfection.
One way to reduce the load of pathogens in the environment is to pay attention to stocking density. We usually think of managing stocking density for group pens, but the same concept can be applied in individual calf housing. By spacing out hutches we may be able to change the dynamics of the pathogens in the environment, creating more open space for sun and wind to affect pathogen environmental survival. Although this seems a standard management tool, there is no data to support the effectiveness of this intervention.
Sanitation and hygiene are the stalwart practices for pathogen reduction. Yet, in farm situations, with the exception of the milking system because of fluid milk handling for human consumption, we do not have specific, evidence-based protocols on cleaning and disinfection. This is true for cleaning the calf nursery area. Almost every extension publication and dairy magazine article that discusses calf diseases talks about biosecurity, sanitation and hygiene, yet there are only a few studies that demonstrate the effect of these practices. In a large study of Norwegian dairy calves, a number of risk factors were found to increase the prevalence of diarrhea in neonatal calves (Gulliksen et al., 2009). If the bedding was removed weekly from calves in individual pens, the risk for diarrhea was reduced four-fold. In a study of salmonellosis in calves, transport vehicles and sales yards were evaluated (Wray et al., 1991). Salmonella were isolated from floors or walls from half of the 14 sales yards. Before washing, Salmonella were found in 20 percent of 107 samples taken from transport vehicles and 6.5 percent of vehicles after washing (no details on cleaning procedures were given). In a study of Salmonella in calf holding areas, the bacterium was found even after housing areas were cleaned suggesting that cleaning was not always effective (Wray et al., 1990b). Again, it is intuitive that cleaning may help decrease environmental loads of pathogens, but there is little or no data to describe what is the proper approach to truly “clean up” the calf’s environment – i.e. reducing the pathogen load.
As mentioned previously, water and feeds can be sources of infection and also become sufficiently contaminated as to present infectious doses to calves. Water was a persistent source of E coli O157 in calves housed in superhutches on one farm in Wisconsin (Shere et al., 1998). Others have shown the same effect for Salmonella (Kirk et al., 2002a). There is no literature to support how to keep water troughs clean and some literature suggests that a stable water trough flora (not cleaned) may reduce bacteria like E coli O157:H7 (FDIU:CVM: WSU unpublished data).
Putting it all together – Protocols for cleaning and monitoring
One farm-specific, bottoms-up method of reducing calfhood disease through decreasing the environmental load is to develop a Hazard Analysis Critical Control Point (HACCP) program for the calf-rearing area. This process helps in developing the protocols and monitoring steps necessary for reducing any kind of hazard – in this case, pathogen reduction in the calf environment (Cullor,
1995; Noordhuizen & Metz, 2005). These programs have had success on dairy farms, particularly with reduction of clinical mastitis and milk quality problems but have not been implemented to any great degree for calf-rearing (Sischo et al., 1995).
Pulling the evidence together, one must consider the timing of clinical cases and pathogen shedding as well as the environmental risk factors for disease when developing protocols that will reduce exposure to pathogens. We have discussed the different hazards – the agents of disease – which have similar points of control, particularly for those that are transmitted through the fecaloral route and result in diarrhea or septicemia in neonatal calves. Where are those points of control? Anything that a calf can touch and anything that touches a calf are potential risks for disease transmission. The following is an example of a list of potential risk items, but the list may be different from farm to farm:
- Other calves
- Calf blankets
- Walls, floors, and roofs of hutches or pens
- Feeding equipment – Mixing tank, hoses, bottles, nipples, buckets
- Treatment equipment – esophageal feeders, balling guns, halters
- Caretakers – hands, boots, clothing
Whatever the item is…attention to cleaning and disinfection will reduce the dose of infectious agents and the risk for disease. Understanding the difference between cleaning and disinfection will help you advise others on how to do the job right. There is quite a bit of information available about cleaning and disinfection. Establishing the farm-specific protocol and then monitoring the behaviors that make up that protocol is the more challenging part. Monitoring cleanliness of bottles and surfaces through swabbing and culturing is one method of monitoring outcomes. Calf hygiene scoring might be another way to provide objective information but for both of these methods of monitoring, providing feedback to caretakers is the only way to improve compliance.
When formulating cleaning protocols, caretakers need to know about the kinds of pathogens, how they spread to calves, and what tools they have to help reduce pathogen loads. Cleaning agents are used to remove dirt, scum, or manure. Some are soaps, and may not include disinfecting agents. Their primary function is to release the potential infecting material from the fomite and reduce the amount of organic material that may interfere with the action of the disinfectant. It is important to CLEAN a surface first BEFORE using a DISINFECTANT (Moore, 2004). For hutches or pen walls/floors, the order would be: 1) scrape (remove the big particles), 2) wash (with a soap or detergent to “lift” dirt and microbes), 3) rinse, 4) disinfect, 5) rinse, and 6) dry. Drying or allowing sun exposure (UV light irradiation) on surfaces is an important step. If using a disinfectant, the contact time, concentration, temperature, pH, hardness of the water, and amount of organic material present determine the success of chemical disinfection (Barrington et al., 2002). Disinfectant labels provide all the needed information on their use. For information on reading labels, see Iowa State University’s Center for Food Security and Public Health

Cleaning steps for feeding equipment would include:
- rinse with lukewarm water
- use detergent and wash
- rinse detergent and soil loosened by it, and
- complete the disinfection step
Cleaning panels within pens and any equipment that might be shared between calves would have similar steps.
If the farm is using outdoor hutches, when a calf is moved, the hutch should be tipped up and scraped of all manure from its surfaces and the pack underneath. Hutches then should be cleaned and disinfected and allowed to stand upright in the sun for several days (if very hot) or up to a week before moving in another calf. It would be ideal to move the hutches to a new location. Some producers may be tempted to refill a hutch if a calf dies. The hutch should not be refilled because of the potential the calf was shedding large numbers of pathogens. The empty hutch also is a way to estimate mortality on a large calf raising operation.
Commonly recommended for disease control in calves is to work from youngest to oldest; but there is no research on this. If we look at the chart of diarrhea prevalence, the highest shedding period is about 2 to 3 weeks of age, when the prevalence of diarrhea is highest. Working with these calves and then younger ones is likely riskiest. Calf caretakers who contact feces or saliva, should disinfect hands and boots and perhaps change clothing before touching another calf. As much work as possible should be done outside the hutch and caretakers should be discouraged from going in and out of hutches.
Summary
Reducing the load of pathogens in the calf environment involves improving host immunity to limit infection and shedding, paying attention to cleaning and disinfection of materials within the calf’s environment, and preventing new pathogen introduction through separation of young calves from each other and from older animals. Caretakers should understand what the hazards are and why protocols for cleaning and disinfection are needed and should get feedback from monitoring for compliance. Reduction of the pathogen load in the calf environment will reduce the burden of illness on the calves, as well as reduce the potential for pathogens to reach the human population.
References
- Alexander, K. A., Warnick, L. D., Cripps, C. J., McDonough, P. L., Grohn, Y. T., Wiedmann, M. et al. (2009). Fecal shedding of, antimicrobial resistance in, and serologic response to Salmonella typhimurium in dairy calves. J Am Vet Med Assoc, 235, 739-748.
- Barrington, G. M., Gay, J. M., & Evermann, J. F. (2002). Biosecurity fo neonatal gastrointestinal diseases. Vet Clin Food Anim, 18, 7-34.
- Bartels, C., Holzhauer, M., Jorritsma, R., Swart, W., & Lam, T. (2010). Prevalence, prediction and risk factors of enteropathogens in normal and non-normal faeces of young Dutch diary calves. Preventive Veterinary Medicine, 93, 162-169.
- Cullor, J. S. (1995). Implementing the HACCP program on your client’s dairies. Veterinary Medicine, 90, 290-295.
- Cummings, K. J., Warnick, L. D., Alexander, K. A., Cripps, C. J., Grohn, Y. T., James, K. L. et al. (2009). The duration of fecal Salmonella shedding following clinical disease among dairy cattle in northeastern USA. Prev Vet Med, 92, 134-139.
- Shere, J. A., Bartlett, P. C., & Kaspar, C. W. (1998). Longitudinal study of Escherichia coli dissemination on four dairy farms in Wisconsin. Appl Env Micro, 64, 1390-1399.
- Sischo, W. M., Hutchinson, L. J., Reneau, J. K., & Timms, L. L. (1995). Total quality management for dairy herds. In (pp. 222-224). National Mastitis Council.
- Smith, D. R., Frotelueschen, D., Knott, T., & Ensley, S. (2003). Managing the alleviate calf scours: the sandhills calving system. In.
- Svensson, C., Lundborg, K., Emanuelson, U., & Olsson, S.-O. (2003). Morbidity in Swedish dairy calves from birth to 90 days of age and indivdual calf-level risk factors for infectious diseases. Preventive Veterinary Medicine, 58, 179-197.
- Trotz-William, L., Martin, S. W., Leslie, K., Duffield, T., Nydam, D. V., & Peregrine, A. S. (2008). Association between management practices and within-herd prevalence of cryptosporidium parvum shedding on dairy farms in southern Ontario. Preventive Veterinary Medicine, 83, 11-23.
- Trotz-Williams, L. A., Martin, S. W., Leslie, K. E., Duffield, T., Nydam, D. V., & Peregrine, A. S. (2007). Calf-level risk factors for neonatal diarrhea and shedding of Cryptosporidium parvum in Ontario dairy calves. Prev Vet Med, 82, 12-28.
- Uga, S., Matsuo, J., Kono, E., Kimura, K., Inoue, M., Rai, S. K. et al. (2000). Prevalence of Cryptosporidium parvum infection and pattern of oocyst shedding in calves in Japan. Vet Parasit, 84, 27-32.
- Uhde, F. L., Kaufmann, T., Sager, H., Albini, S., Zanoni, R., Schelling, E. et al. (2008). Prevalence of four enteropathogens in the faeces of young diarrhoeic dairy calves in Switzerland. The Veterinary Record, 163, 362-366.
- Veling, J., Wilpshaar, H., Frankena, K., Bartels, C., & Barkema, H. W. (2002). Risk factors for clinical Salmonella enterica subsp. enterica serovar Typhimurium infection on Dutch dairy farms. Prev Vet Med, 54, 157-168.
- Wray, C., Todd, N., McLaren, I., Beedell, Y., & Rowe, B. (1990a). The epidemiology of Salmonella infection of calves: the role of dealers. Epidemiol.Infect., 105, 295-305.
- Wray, C., Todd, N., McLaren, I., Beedell, Y., & Rowe, B. (1990b). The epidemiology of salmonella infection of calves: the role of dealers. Epidemiol Infect, 105, 295-305.
- Wray, C., Todd, N., MsLaren, I. M., & Beedell, Y. E. (1991). The epidemiology of salmonella in calves: the role of markets and vehicles. Epidemiol Infect, 107, 521-525.
- Younis, E. E., Ahmed, A. M., El-Khodery, S. A., Osman, S. A., & El-Naker, Y. F. I. (2009). Molecular screening and risk factors of enterotoxigenic Escherichia coli and Salmonella spp. in diarrheic neonatal calves in Egypt. Research in Veterinary Science, 87, 373-379.